Conference Report
ISPAD 2024 — Seven Takeaways from Lisbon
ISPAD 2024 in Lisbon highlighted the extraordinary pace of research into type 1 diabetes screening and prevention, shone a spotlight on type 2 diabetes in children, and showed promising early results for GLP-1 therapies in paediatric diabetes.
Overview
This report covers seven key themes from ISPAD 2024 — the International Society for Paediatric and Adolescent Diabetes annual conference.
This content is for educational exploration only. It describes average responses and general principles. It is not medical advice and cannot replace individual clinical guidance from your diabetes care team.
1. Delaying Progression to T1D Stage 3
The ADA consensus guidelines recommend early identification of T1D stages 1 and 2 as a critical window for preparation — reducing DKA risk, developing glucose management skills, and avoiding hospitalisation. The guidelines recommend monitoring glucose regulation every three months, measuring random venous or capillary blood glucose alongside HbA1c, using OGTT to assess T1D staging, and referring to specialists in T1D progression wherever feasible.
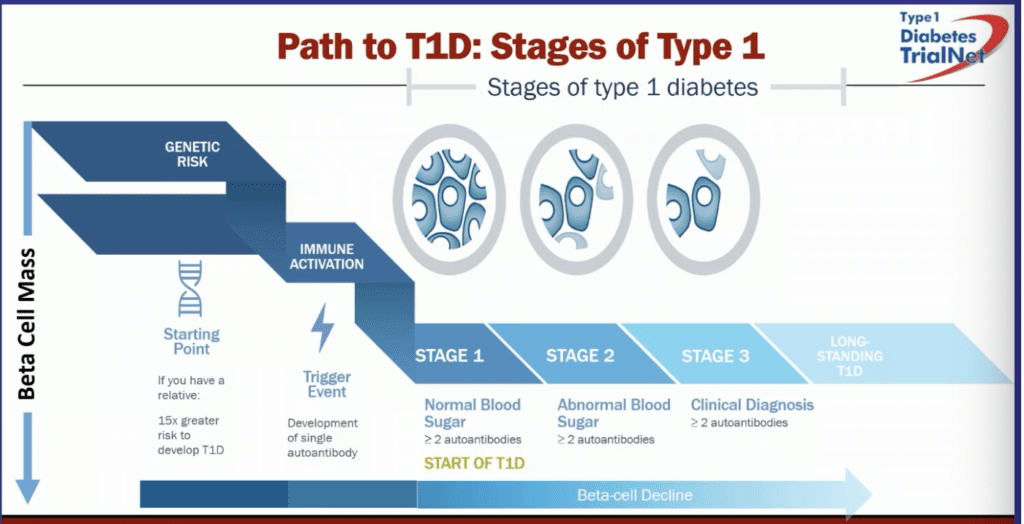
The Fr1da study showed that 90% of people identified with stage 2 T1D did not have a first-degree relative with the condition — highlighting the importance of population-level screening, not just family-based programmes. Teplizumab (Tzield) is licensed for children aged 8 and above after showing a 2–3 year delay in progression to T1D stage 3. It is currently undergoing NICE appraisal. Trials in younger children (aged 2–8) are under way.
Colin Dayan from Cardiff discussed the multiple immune-modulating agents being tested, and envisaged a future where combinations of agents might achieve much longer delays. C-peptide preservation needs to be recognised as a regulatory-accepted endpoint for this work to accelerate. The T1D Plus platform format will allow multiple agents to be tested using a single control group, accelerating the science.
Dr. Tabitha Randell discussed preservation approaches including infusion therapies (teplizumab, ATG, rituximab, abatacept, alefacept, golimumab), subcutaneous injections (ustekinumab, imatinib), and oral medications (baricitinib, verapamil). Key challenges include identifying who to screen, selecting which autoantibodies to test and when, determining the most effective screening ages (likely 2–4 and 6–8 years), integrating screening into routine practice, and identifying candidates for novel treatments.
2. AID from Diagnosis, or Within Three Months
For the vast majority of children newly diagnosed with type 1 diabetes, starting an automated insulin delivery (AID) system earlier is associated with better glucose management outcomes at both one and two years.
The CLOUD trial compared AID systems with multiple daily injections (MDI) in over 100 newly diagnosed children in a randomised controlled trial. AID users achieved 73% Time in Range (TIR) compared to 60% for those on standard therapy, with reduced glycaemic variability and insulin dosing errors, and lower hypoglycaemia rates.
The significance of early glycaemic management relates to the long-term evidence from the DCCT and EDIC studies, which show that better glucose management in the early years of type 1 diabetes reduces the long-term risk of both microvascular and macrovascular complications.
3. Metformin and Lifestyle for Youth-Onset T2D Will Not Be Enough
The TODAY and TODAY2 studies together provide a longitudinal picture of youth-onset type 2 diabetes and the scale of the challenge.
TODAY Study (2004–2012)
In 699 participants aged 10–17 with type 2 diabetes, the treatment failure rate in maintaining glycaemic management was high: 51.7% with metformin alone, 46.6% with metformin plus lifestyle, and 38.6% with metformin plus rosiglitazone. Youth experienced faster disease progression than is typically seen in adults.
TODAY2 Study (2012–2020)
By a mean age of 26 years, participants showed high rates of complications: hypertension in 67.5%, diabetic kidney disease in 54.8%, neuropathy in 32.4%, and retinopathy in 51%. Multiple participants developed cardiovascular complications including heart failure. The majority had at least one major complication; 28.4% had two or more.
Data from the German DPV registry, presented by Dr. Dezer, showed even higher rates of micro and macrovascular complications in youth living with type 2 diabetes than those seen in TODAY, underscoring the aggressive progression of the condition in this age group.
The emerging message: early and effective metabolic management — potentially including GLP-1 or SGLT-2 agents — is essential, not optional.
4. Adding GLP-1 Agonists or SGLT-2 Inhibitors in Youth-Onset T2D
The AWARD-PEDS trial (n=154) assessed dulaglutide (a GLP-1 receptor agonist) in children with type 2 diabetes; the DINAMO trial (n=158) explored dapagliflozin (an SGLT-2 inhibitor). Dulaglutide reduced HbA1c by 1.1% over 26 weeks; dapagliflozin reduced HbA1c by approximately 0.8% in adolescents. Both were well tolerated.
In obese young people without type 2 diabetes, a semaglutide trial showed significant weight loss compared to placebo, with potential implications for T2D prevention and remission.
Updated ISPAD guidelines for type 2 diabetes in youth were anticipated, recommending metformin and lifestyle as the foundation, with basal insulin if HbA1c exceeds 69 mmol/mol. The direction of travel is toward adding GLP-1 or SGLT-2 agents early if metformin and lifestyle cannot achieve HbA1c below 48 mmol/mol, and transitioning away from insulin as soon as feasible.
5. Obesity Assessment Using Waist-to-Height Ratio
Research presented at the conference explored the relationship between waist-to-height ratio (WHtR) and insulin resistance in overweight and obese children. Children in the highest WHtR tertile showed significantly higher risks of insulin resistance and elevated liver enzymes. WHtR appears to be a practical screening tool for metabolic syndrome in children, and may serve as a sensitive marker of response to intervention.
WHtR tertiles were divided by sex: males T1 at or below 0.55, T2 above 0.55 to 0.59, T3 above 0.59; females T1 at or below 0.56, T2 above 0.56 to 0.60, T3 above 0.60.
6. Time in Tight Range (TITR) for Well-Managed Patients
A real-world analysis of paediatric AID users in Italy (n=613) focused on TITR — defined as glucose between 3.9 and 7.8 mmol/L (70–140 mg/dL) — as a potential additional metric for those already achieving good TIR. The cohort achieved an average TITR of 47%, with top-quartile users reaching over 50%, without increasing hypoglycaemia risk (time below range remained under 4%).
The clinical suggestion from the conference: aiming for 50% TITR is reasonable if the person is already achieving 70% TIR. Setting TITR as an additional target before 70% TIR is achieved risks creating unrealistic expectations and increasing distress. For most people to achieve 50% TITR, a second agent — such as an SGLT-2 inhibitor, GLP-1 receptor agonist, or amylin analogue — may be needed to address post-meal hyperglycaemia. BREAKTHROUGH T1D is funding several trials in this area.
7. Exercise in Children with T1D and T2D
Children with type 2 diabetes show substantially lower VO2 max and reduced fat oxidation capacity compared to their peers, partly due to insulin resistance. This means moderate-intensity physical activity is often genuinely difficult for them. A low-intensity starting point with gradual progression is generally appropriate. GLP-1 or SGLT-2 agents as the second-line treatment may improve capacity to benefit from physical activity over time.
An upcoming EASD/ISPAD consensus statement on AID systems and exercise in type 1 diabetes — covering general recommendations, device-specific guidance, and specialty circumstances — was anticipated. A summary will be published once the statement is available.
Report by John Pemberton. Shared for informational and educational purposes.
